Product
제품
소개
Proteus NoEndo™ μ
 Residual endotoxin contamination in advanced biotherapy products is an expensive and often difficult contaminant to control.
Residual endotoxin contamination in advanced biotherapy products is an expensive and often difficult contaminant to control.
Many commercially-available protocols are unable to remove endotoxins effectively and are based on non-affinity chromatography methods e.g. Ion exchange chromatography, phase separation using Triton X-114 or require time consuming and expensive affinity steps. These costly resins are often supplied as loose resin or packed in slow gravity columns.
The Proteus NoEndo™ spin column kits offer a standardised method for high grade clearance of endotoxin from recombinant proteins, antibodies and viral vectors.
The SelfSeal™ Advantage 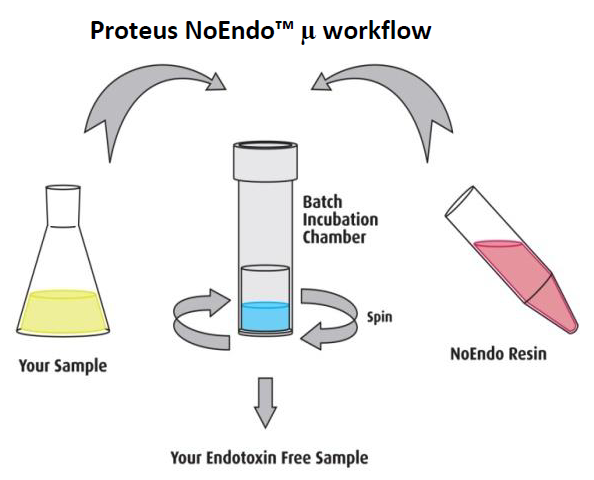
NoEndo™ μ spin columns incorporate our proprietary and NASA-inspired
SelfSeal ™ membrane technology. The membrane is specially formulated to
prevent any sample from leaking into the collection tube on an orbital mixer.
In a centrifuge, the membrane pores dilate and the eluate, free of endotoxin,
passes into the collection tube. The contact time is maximized to ensure
maximum endotoxin depletion without losses of the target protein, antibody or
domain antibody. Uniquely, there is also no dilution of the sample.
사양
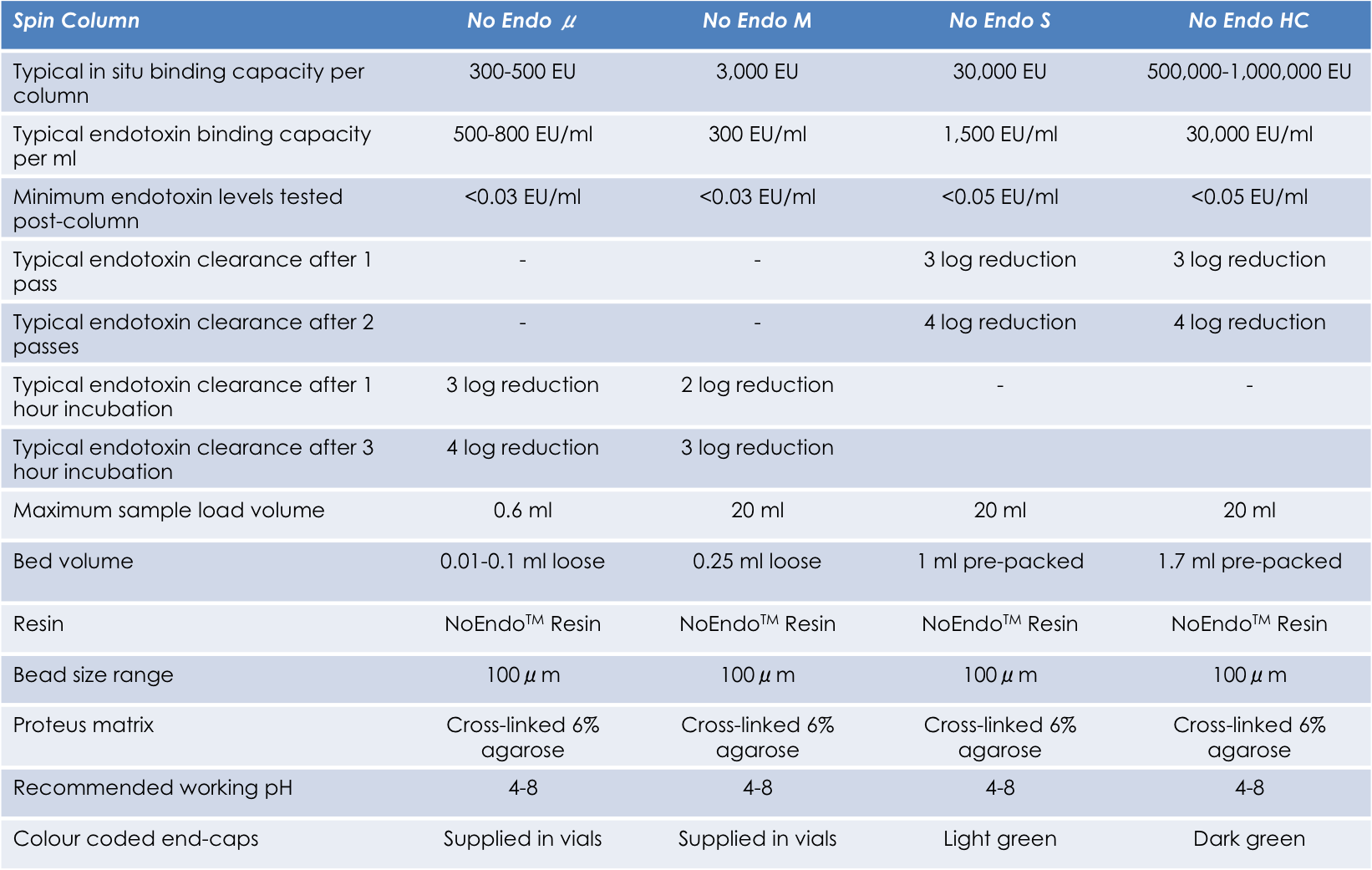
주문정보

관련자료
Proteus NoEndo μ Brochure
Proteus NoEndo μ Protocol
Proteus NoEndo μ Userguide
Proteus NoEndo Resin Userguide

