Product
제품
소개
Proteus NoEndo™ M (Mini), NoEndo™ S (Standard) and
NoEndo™ HC (High Capacity) Spin Column Kits:

Residual endotoxin contamination in advanced biotherapy products is an expensive and often difficult contaminant to control. Many commercially-available protocols are unable to remove endotoxins effectively and are based on non-affinity chromatography methods e.g. Ion exchange chromatography, phase separation using Triton X-114 or require time consuming and expensive affinity steps. These costly resins are often supplied as loose resin or packed in slow gravity columns.
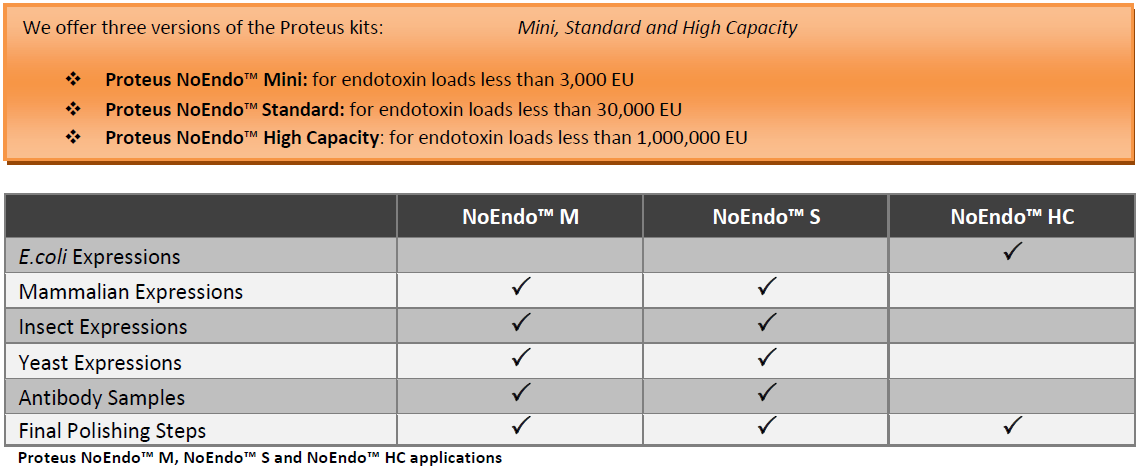
The Proteus NoEndo™ spin column kits offer a standardised method for high grade clearance of endotoxin
from recombinant proteins, antibodies and viral vectors. These agents are increasingly being designed for
therapeutic applications, hence moving them forward efficiently through in vivo studies requires pure preparations of the samples.
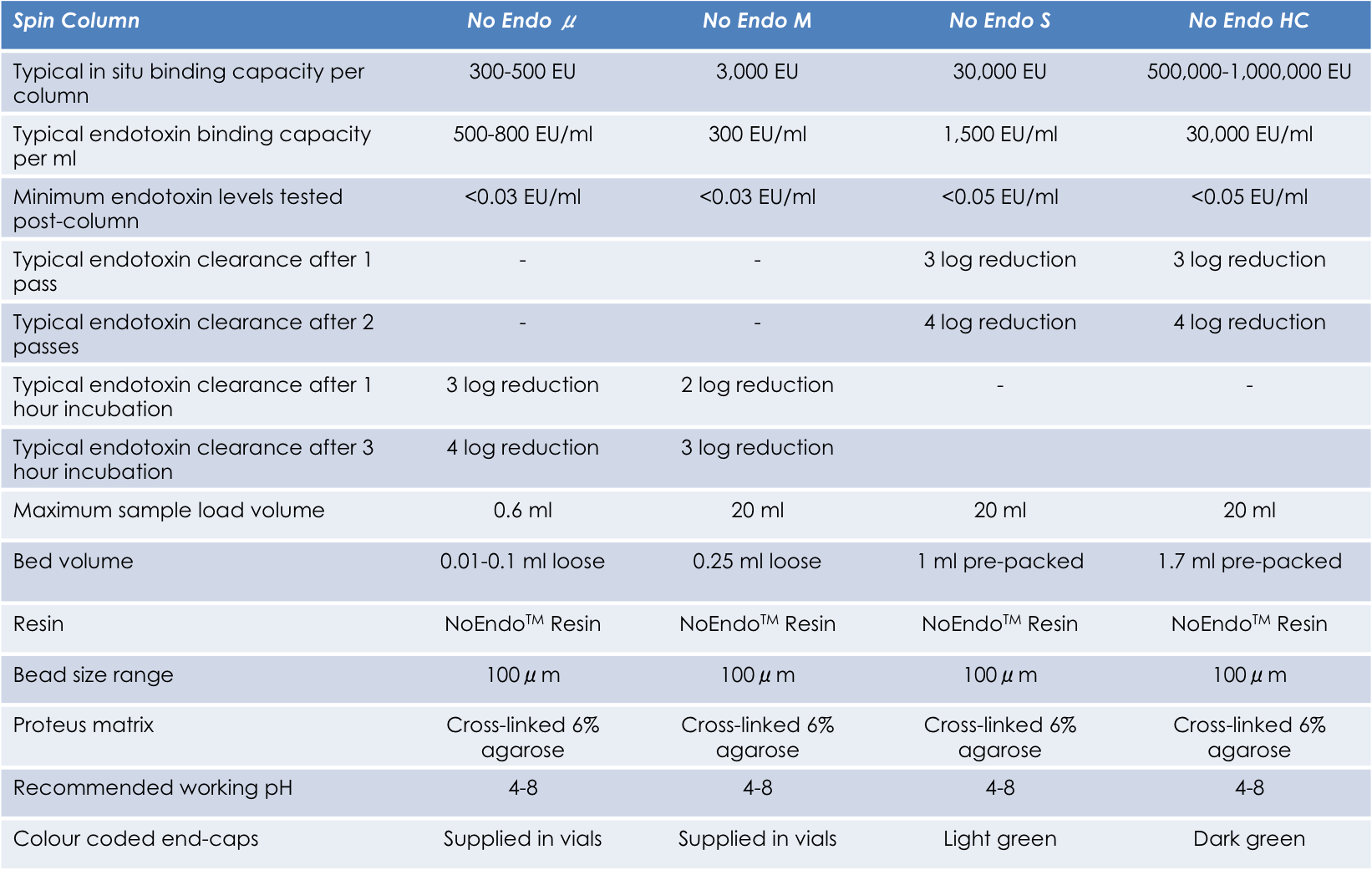
Next generation Proteus kits combine the quality separation you expect from gravity flow columns with the speed andease-of-use of spin columns. Both column formats reveal a high degree of innovation! The Standard and High Capacitycolumns incorporate pre-packed resin cartridges utilizing our FlowGo™ technology. The Mini columns are emptycolumns that incorporate our proprietary SelfSeal™ membrane technology. This ensures that there is no passage ofthe sample through the membrane during the batch incubation at 40C or at room temperature.
사양
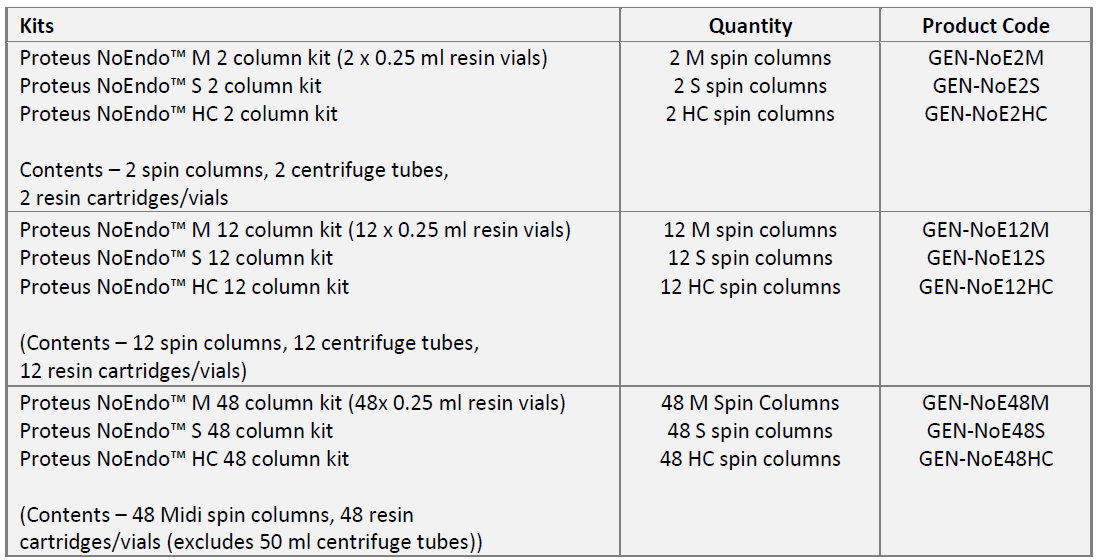
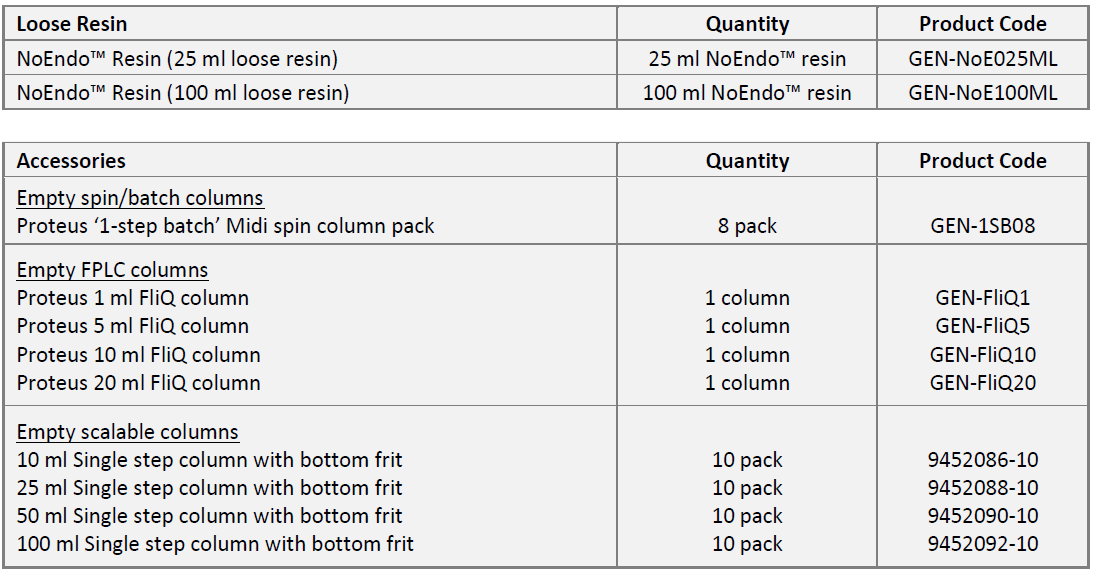
주문정보
관련자료
Proteus NoEndo M, S and HC Brochure
Proteus NoEndo M Protocol
Proteus NoEndo M Userguide
Proteus NoEndo S and HC Protocol
Proteus NoEndo S and HC Userguide
Proteus NoEndo Resin Userguide

