Solutions
솔루션
광산란
고분자 및 나노입자의 몰 질량, 크기, 전하/제타전위, 상호작용, 형태 및 접합체 분석 home
home- > 솔루션 >
- 광산란
|
Solutions for Vaccine Characterization and Development
인성크로마텍(주)
|
| Date : 2020.04.10 |
| 분류 : 바이오테라퓨틱스 |
To ensure safe and efficacious vaccines, it is essential to implement robust and reliable characterization tools across discovery, development, production and quality control.
Wyatt’s suite of solutions analyzes critical vaccine attributes, including:
• Molar mass and size • Titer and nucleic acid content • Aggregation • Thermal and colloidal stability
Many of these assays can be streamlined and automated for high throughput.
Our technologies are also helpful in vaccine research, for studying the binding affinity and stoichiometry of antibody-antigen complexes, the oligomeric state of surface glycoproteins, or optimizing formulation conditions.
Continue reading to learn how Wyatt products are applied to characterization of specific vaccine classes.
PL9006: Key publications on the characterization of vaccines with multi-angle and dynamic light scattering
mRNA / DNA
Vaccines based on mRNA or DNA are often delivered via lipid nanoparticles, virus-like particles or other nanoparticle vectors. Multi-angle and dynamic light scattering (MALS, DLS) are useful in characterizing the mRNA itself, the carrier particle, and the complete vaccine.
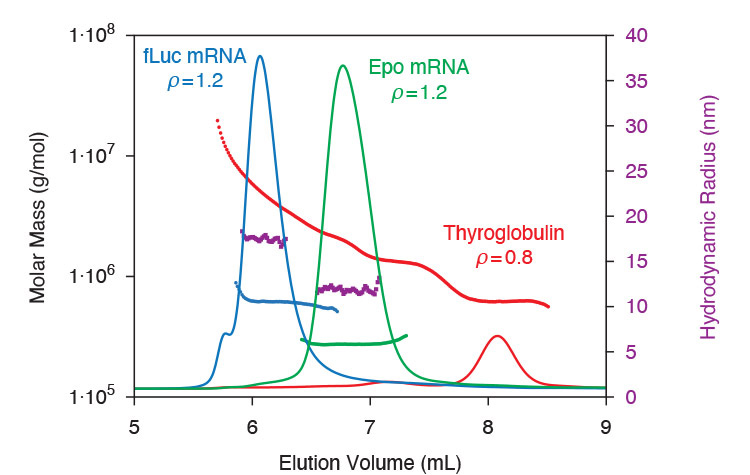
mRNA biophysical attributes As described in application note AN1616: SEC-MALS methods for characterizing mRNA, key biophysical attributes of mRNA are quantified by SEC-MALS plus DLS. This analysis determines molecular weight, percentage of aggregates, radius of gyration Rg, and hydrodynamic radius Rh. The relationship between MW and Rg identifies single- or double-stranded RNA, while the ratio Rg:Rh indicates molecular conformation.
This method can be readily adopted to further study how these attributes are affected by mRNA preparation, formulation, and
storage conditions.
SEC-MALS-DLS determines correct molecular weight and size values for mRNA, even though the molecules elute quite differently than globular protein standards such as thyroglobulin.
Characterization of LNP delivery vehicles Lipid nanoparticles (LNPs) include nano-emulsions, micelles, and solid lipidnanoparticles. One of the mostimportant parameters for uptake in the lymph node, to target the antigen-presenting cells, is size. Only LNPs smaller than about 150 nm in diameter are taken up, while larger LNPsare retained at the injection site. This presents a narrow window of size forthe best-performing LNPs.
FFF-MALS determines high-resolution size distributions, quantifying particleconcentration at each size fraction, in order to qualify precisely the efficacyof a LNP mRNA carrier. Wyatt Technology provides turn-keyFFF-MALS systems based on the Eclipse™ FFF controller and DAWN® multi-angle lightscattering detector, with comprehensive software for method development, instrument control, data acquisition and analysis. For more details on the applications of FFF-MALS in LNP characterization, see WP2608: Lipid nanoparticle and liposome characterization with FFF-MALS.
A complete SEC-MALS system comprises the HPLC including the UV detector, SEC column(s), MALS and RI detectors. Control, data acquisition and analysis are performed by ASTRA® software.
LNP process analytics Once a successful LNP/mRNA platform has been developed, scaling up the process is facilitated by real-time multi-angle light scattering (RT-MALS). With ultraDAWN™ either in-line or on-line, continuous monitoring of critical quality attributes like size and molar mass is achieved. Real-time monitoring of liposome size in a production environment is described in AN8006: In
line monitoring of liposome size by RT-MALS.
Viruses (inactivated or live-attenuated)
These complex vaccine products combine multiple serotypes, often with adjuvants. Asymmetric-flow field-flow fractionation (AF4) separates such complex samples by size with high resolution. Fractionation is followed downstream by multiple online instruments, including MALS, DLS, UV and fluorescence detectors, to complete the biophysical characterization.
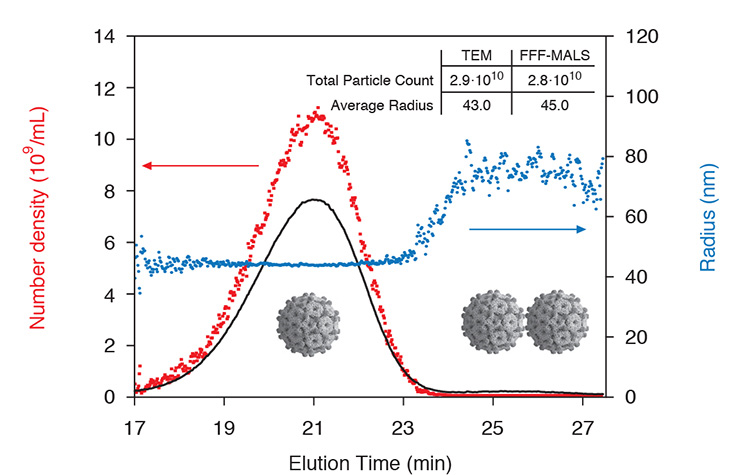
Product validation In the analysis of an influenza virus subpopulation shown here, both size and total particle concentration are quantified in detail by FFF-MALS. Comparison with results from TEM shows excellent agreement within 2% in virus concentration and 5% in size. A small dimer peak is also identified.
Complementary to FFF-MALS, the DynaPro® Plate Reader provides high-throughput screening of hundreds of samples with respect to processing conditions and formulations. In addition to sizing, this technique is able to screen for thermal stability of virus samples and to determine viral particle concentration.
Additional literature See http://doi.org/10.1016/J.JVIROMET.2007.04.008 to explore the comparison of FFF-MALS analysis to other methods such as transmission electron microscopy (TEM), atomic force microscopy (AFM), size exclusion chromatography followed by MALS (SEC-MALS), quantitative reverse transcription polymerase chain reaction (RT Q-PCR), median tissue culture dose (TCID50), and the fluorescent focus assay (FFA).
FFF-MALS is an excellent tool for vaccine characterization. Total viral count and size may be quantified with high resolution for each subpopulation.
Virus-like particles (VLPs)
Recombinant virus-like particles, which are designed to elicit an immune response in the same way that a traditional live- attenuated vaccine does, are typically comprised of a protein capsid, with nucleic acid cargo. SEC-MALS and FFF-MALS characterize both the capsid and cargo, while DLS is suitable as a quality-control tool to determine size, aggregation and titer.
Monitoring production process: measuring protein capsid and DNA cargoThe synthesis of VLPs involves three steps: 1) isolation of recombinant VLP from cell culture, 2) dissociation of the virus into pentameric capsomeres with removal of all DNA, 3) introduction of the DNA of interest and packaging into a newly formed VLP.
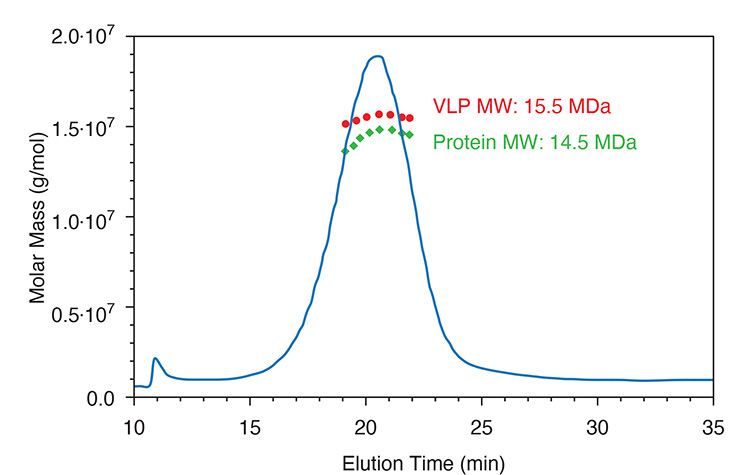
FFF-MALS is used to monitor this process, by separating and detecting fully formed capsids from aggregates, unassembled protomers, and capsomeres. The analysis informs on conformation, and, in the final step of the process, simultaneously measures protein and DNA molar mass to quantify the payload.
Additional literature PL2609: FFF-MALS-DLS nanoDDS key publications contains references to publications highlighting the use of FFF-MALS to characterize liposomes, polymersomes, virus-like particles and other potential gene vectors delivery vehicles.
View the webinar VLP Characterization: The Light Scattering Biophysical Toolbox for additional means of characterizing VLPs and their interactions with neutralizing antibodies.
ASTRA software simultaneously determines molar mass distribution for VLP and protein to quantify DNA payload.
See http://doi.org/10.1016/j.bej.2015.04.004 to learn about a rapid and simple screening method, based on high-throughput dynamic light scattering using the DynaPro Plate Reader, used to identify conditions for enhanced stability of modular vaccine candidates consisting of capsomeres.
The DynaPro Plate Reader enables rapid screening of formulations for size and stability.
Polysaccharide and glycoconjugate vaccines
Polysaccharide and protein glycoconjugate vaccines combine adjuvants with high-molecular-weight polysaccharide antigens, typically including several serotypes. In protein glycoconjugates, protein adjuvants are covalently linked directly to the polysaccharide.
Since molecular weight is central to the immunogenicity of this type of vaccine, SEC-MALS and FFF-MALS are essential for their characterization and quality control.
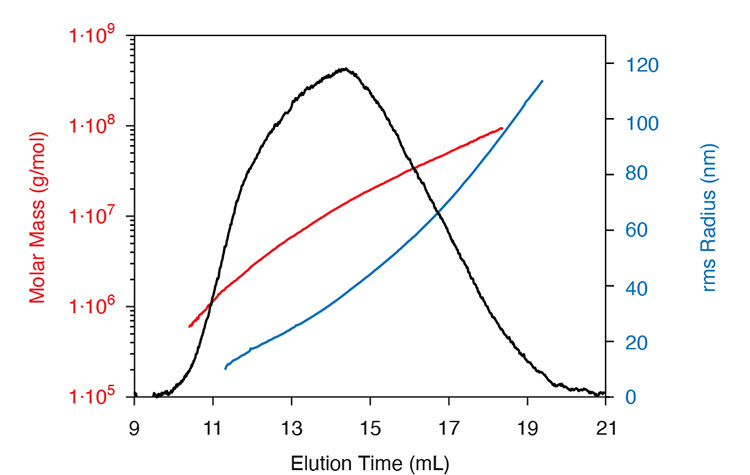
Size and molar mass characterization FFF‐MALS, combining an Eclipse FFF controller, DAWN MALS detector, Optilab® dRI detector and industry-standard HPLC components, is the preferred means of characterizing protein-polysaccharide conjugate vaccines that extend above tens or hundreds of MDa in MW. As there is no column matrix, there is no shear‐induced degradation while fractionating the entire range; MALS determines molar mass and size simultaneously.
For smaller molecules, SEC-MALS may be more readily utilized. Typically, SEC-MALS can determine not only the overall molar mass of the molecule, but also quantify the content of protein and content of polysaccharide in each eluting fraction. A SEC-MALS system includes HPLC pump, autosampler and column, a DAWN and Optilab, and may also include a ViscoStar® differential viscometer for additional analysis of conformation.
More details on the analysis of protein-polysaccharide conjugate vaccines and other vaccine types are explained in our webinar Vaccines, Well Characterized? Light scattering solutions for biophysical characterization in vaccine R&D.
FFF-MALS precisely characterizes molar mass and size distributions for large protein-polysaccharide conjugates.
Additional literature AN8005: RT-MALS end-point determination of a polysaccharide depolymerization process describes how the ultraDAWN real-time MALS detector monitors polysaccharide molecular weight during processing to ensure optimalquality on the production floor.
AN1306: Polyvalent pneumococcal polysaccharide vaccine characterized by SEC-MALS describes how MALS can be used to aid quality control in vaccine QC.
Protein antigens
Protein vaccines are a type of subunit vaccine, typically a surface antigen. As
with monoclonal antibodies and other therapeutic proteins, DLS and SEC-MALS are invaluable tools for characterization, formulation and process development of protein antigen products.
Monitoring of purification of a protein vaccine from inclusion bodies with DLS In industrial-scale production of a recombinant fusion protein vaccine, the process of solubilizing inclusion bodies in urea and refolding is monitored by DLS. In this study, it could be demonstrated that disulfide bonds prevent formation of folded monomers from the inclusion bodies, and the refolding should be carried out in a reducing environment. See https://doi.org/10.1002/jps.23746 to learn more about this study.
Validation of trimeric surface glycoproteinsIdeally an antigen utilized in a vaccine against COVID-19, or a target for a therapeutic antibody or small molecule drug, would incorporate a fully trimeric nCOV-19 spike glycoprotein. SEC-MALS is the technique of choice to confirm the oligomeric state of a glycoprotein in solution, and is used by providers of these reagents to validate their products. In addition to absolute molar mass and oligomeric state, SEC-MALS also determines the degree of glycosylation of such glycoproteins.
Explore further
Bibliography MALS, DLS and FFF are used extensively in structural virology and the study of viral proteins. A large selection of peer-reviewed publications in these fields that cite measurements obtained with Wyatt instruments may be found by searching our Bibliography. For example, a search on the keyword “coronavirus” returns 10 results related to SARS, MERS and other coronaviruses, while a search on the keyword “influenza” returns over 60 results.
Highlight on SARS-CoV In an effort to understand the structure and function of nsp1, a relevant therapeutic target of SARS-CoV, SEC-MALS using the miniDAWN® and Optilab, was critical to unambiguously establish the oligomeric state as a monomer while traditional SEC indicated a dimer. Read more about this study here: http://doi.org/10.1016/j.pep.2006.11.005.
Discover our solutions
Download our latest brochure which highlights tools and techniques for characterizing biologics and complex drugs:
|